These means learners must divide their entropy by 1000 before using it in the Gibbs equation.Īnother common mistake is to use the temperature in ☌ rather than converting to K, so using 25☌ rather than 298 K.Source : Born – Haber cycle- Question 1) – Calculate the lattice enthalpy of KCl from the following data at standard states- Enthalpy of sublimation of K = 89 KJ/mole Enthalpy of dissociation of chlorine = 244 KJ/ mole Ionisation energy of K = 425 KJ/mole Electron gain enthalpy of chlorine = -355 KJ/mole Enthalpy of formation of KCl = -438 KJ/mole Solution )
#Born haber cycle problems with solutions free
Forgetting these in equations relating to enthalpy changes can cost marks, so make a big deal of them.Ī common error when calculating free energy using \(\displaystyle \Delta G = \Delta H - T\Delta S\) is that learners forget that the units for entropy (\(\displaystyle \Delta S\)) are J K –1 mol –1 whereas enthalpy (\(\displaystyle \Delta H\)) is given in kJ mol –1. State symbols are a regular tripping point for learners.

This causes them to add rather than subtract the value and calculate the lattice enthalpy incorrectly. For example, when completing a Born–Haber cycle for MgC l 2, the enthalpy change of atomisation (\(\displaystyle \Delta_aH\)) for chlorine isīut many learners forget that this applies to half a mole of chlorine and would not double it for a whole molecule.Īnother common mistake in a cycle would be that learners forget that \(\displaystyle \Delta_ H^\ominus\) is endothermic (arrow points up). Gibbs energy is a lot harder to visualise and is better explained using the equation, practically the enthalpy and entropy of a system like the one mentioned above can be measured, compared and shown to be feasible.Ĭommon misconceptions or difficulties learners may haveĪ common mistake in Born–Haber cycles is that learners forget to multiply or divide standard values.

Learners grasp that solids are more ordered than gases and the associated entropy change can then be calculated from standard values. The negative values can cause confusion drawing a cycle to scale (see Learner Resource 1) can help learners visualise the cycle before they start calculating it.Įntropy can be easily introduced with some high entropy reactions, for example the reaction of a carbonate and acid. A good method is to include a competitive edge: have pairs write down your choice of equation and element and score points by spotting each other’s mistakes.īorn–Haber cycles contain endothermic reactions like atomisation and exothermic ones like electron affinity. This can be a very dry lesson if just written down and discussed. These are: formation, atomisation, 1 st and 2 nd ionisation, electron affinities and lattice enthalpy.
#Born haber cycle problems with solutions how to
To explain and build Born–Haber cycles you must first have a clear understanding of each of the individual standard reactions and how to define them.
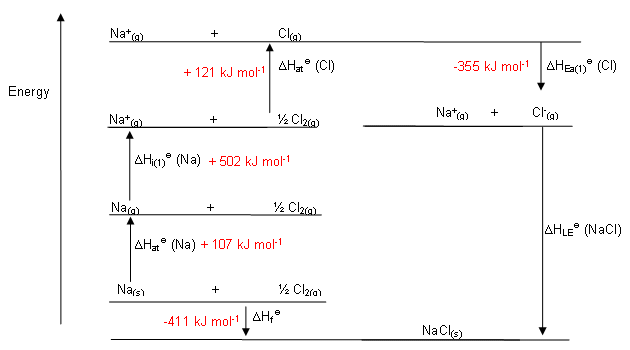
However, the ideas behind entropy can be difficult to visualise and explain and there are many pitfalls which catch out even the brightest of learners. This can lead to a large variety of errors within questions and makes it a very polarising topic.Įntropy and free energy, when boiled down, are fairly straightforward calculations and questions can be attempted with little prior knowledge. The individual reactions involved – such as atomisation, ionisation and electron affinity – can be hard to visualise and their details easy to mix up and forget.

Lattice Enthalpy builds on the concepts learned in Hess cycles and relies heavily on mathematical understanding. This guide focuses on two key areas of A level Chemistry: Lattice Enthalpy and Entropy.
